
1. Executive Summary
PFAS-Free sugarcane tableware is now a procurement threshold, not just a sustainability label. In real-world molded fiber sourcing, the key technical choice is no longer whether to remove fluorinated chemistry, but which fluorine-free grease resistance pathway can still deliver stable performance, scalable manufacturing, and defensible compliance. For most hot takeaway and foodservice scenarios, high-performance internal additive systems offer the strongest balance of grease resistance, process efficiency, and commercial viability. Low-temperature internal additive systems remain relevant for cold-chain, ambient, and low-grease applications, but their limits must be clearly understood. By contrast, physical barrier systems such as lamination and surface coatings may achieve higher barrier performance in niche cases, yet they also introduce higher cost, secondary processing, and more complex repulpable and regulatory considerations. This article provides a technical sourcing framework for evaluating PFAS-Free bagasse tableware technologies across compliance, grease resistance performance, manufacturing scalability, and supplier verification risk, informed by the sourcing logic, manufacturing observations, and quality-control experience developed through long-term molded fiber production and export practice at Bioleader®.
- Key Regulatory Baseline: PFAS-free sourcing now requires verifiable fluorine testing, certification alignment, and market-specific compliance review.
- Mainstream Technical Path: High-performance internal additive systems remain the most practical option for most hot takeaway and foodservice applications.
- Low vs. High Performance: Low-temperature additive systems are suited to cold-chain, ambient, and low-grease foods, while high-performance systems are developed for more demanding takeaway applications.
- Niche Alternative: External barrier systems, including lamination and surface coatings, are mainly justified for special high-barrier requirements.
- Sourcing Priority: Buyers should compare repeatable factory-scale execution, not just laboratory sample performance.
2. Why PFAS-Free Technology Selection Matters in 2026
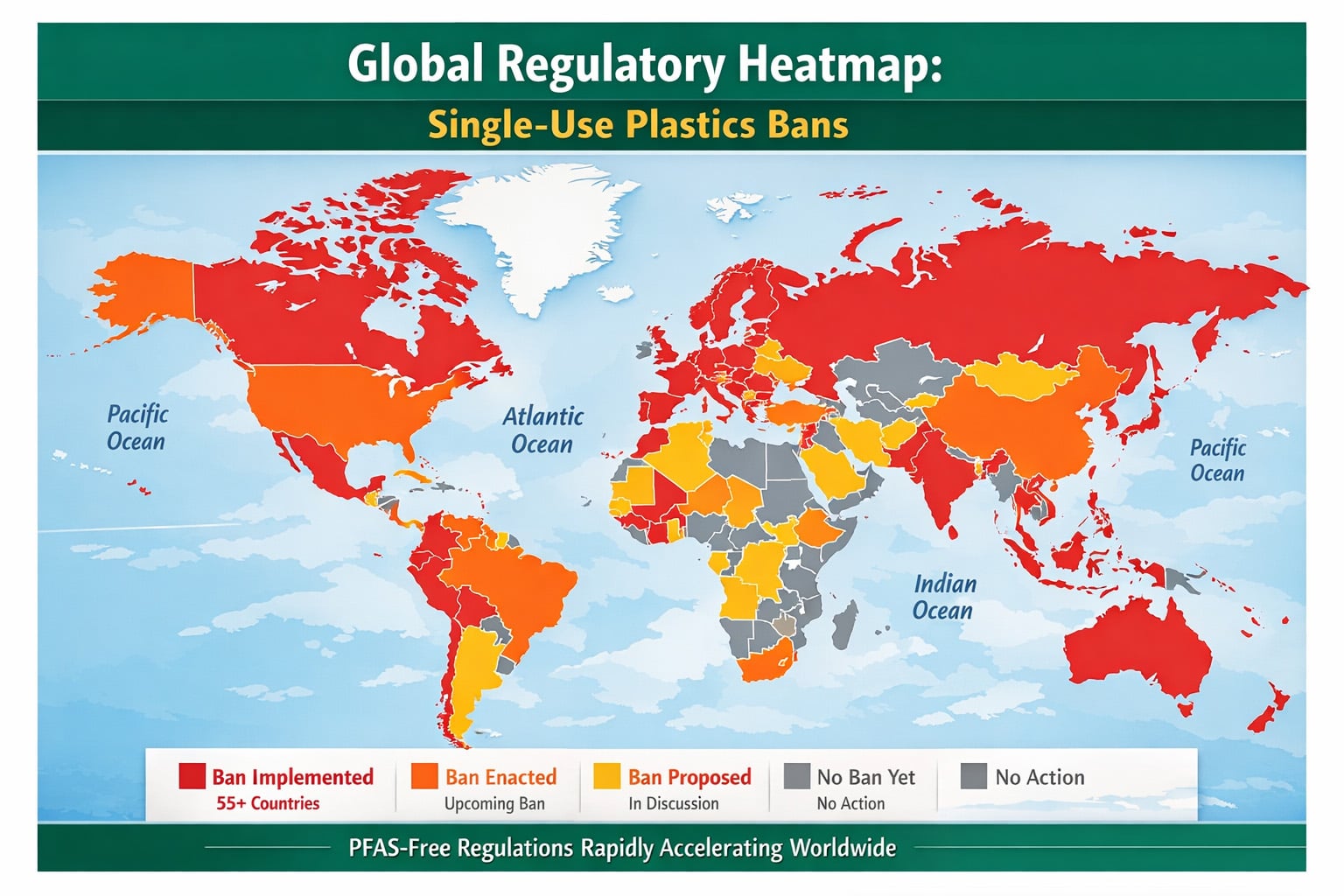
In 2026, PFAS-Free packaging is no longer a branding advantage reserved for sustainability-focused buyers. It has become a baseline sourcing requirement across a growing number of foodservice and food-contact markets. For procurement teams buying molded fiber products such as sugarcane clamshells, plates, bowls, trays, and food containers, the real question is no longer whether fluorinated chemistry should be removed. The real sourcing challenge is which fluorine-free grease-resistance pathway can still deliver repeatable performance, stable production efficiency, and defensible compliance at commercial scale.
Sugarcane tableware now sits at the intersection of three demanding requirements: food-contact safety, grease and moisture resistance, and circular-material expectations. Buyers are no longer comparing a “green” option against a conventional one. They are comparing different PFAS-Free technical routes against each other, each with its own trade-offs in heat tolerance, grease resistance, processing complexity, repulpability, and evidence burden. In practice, this means that material choice alone is no longer enough. The procurement decision must now include barrier design logic, factory execution capability, testing method, and document traceability.
The urgency is especially clear in export-facing supply chains. In the European Union, the new Packaging and Packaging Waste Regulation introduces explicit concentration limits for PFAS in food-contact packaging that apply from August 12, 2026. In North America, buyers are facing a combination of state-level PFAS restrictions, certification-based fluorine screening requirements, and FDA actions that have already removed multiple PFAS-related food-contact notifications from effectiveness. As a result, PFAS-Free claims can no longer rely on vague declarations or marketing language. They must be supported by test logic, product-specific documentation, and sourcing decisions that remain credible under regulatory review.
For molded fiber foodservice packaging, this is why technology selection now matters more than claim language. A low-cost PFAS-Free sample that works for a short ambient test may fail under hot oil, long hold times, or high-fat takeaway use. A high-barrier surface treatment may perform well in isolated trials but become commercially weak once secondary processing, throughput loss, and repulpability pressure are taken into account. The sourcing problem in 2026 is therefore not simply “PFAS-Free or not.” It is how to select the right PFAS-Free pathway for the actual food application, operating temperature, compliance market, and production reality.
This guide is built around that question. It focuses on the two main technical routes now shaping PFAS-Free sugarcane tableware sourcing: Internal PFAS-Free Additive Systems and External Barrier Systems. It also explains why buyers must distinguish between low-temperature and high-performance internal additive technologies, why lamination and coating cannot be treated as interchangeable concepts, and why factory-scale repeatability matters more than one-off sample success. In short, this is not an environmental overview. It is a sourcing framework for making technically and commercially defensible packaging decisions in 2026.
3. Definition Box: Terms, Boundaries, and What “PFAS-Free” Really Means
Before comparing technical pathways, buyers need to clarify the language used in supplier claims, test reports, and product specifications. In PFAS-Free molded fiber sourcing, confusion often begins with terminology. Words such as “PFAS-Free,” “fluorine-free,” “grease-resistant,” “coated,” and “laminated” are frequently used as if they mean the same thing. They do not. For procurement, compliance, and product-development teams, these distinctions are not semantic details. They directly affect how a product is tested, how performance is interpreted, and how a sourcing decision is defended.

| Term | Working Meaning in This Article |
|---|---|
| PFAS-Free | A claim that must be supported by test logic, documentation, and market-specific compliance review. |
| Fluorine-free | A common trade expression that is related to, but not identical with, a full PFAS compliance conclusion. |
| Internal PFAS-Free Additive Systems | Fluorine-free grease- and water-resistance chemistry introduced into the molded fiber process from within the pulp system. |
| External Barrier Systems | Barrier structures added to the molded fiber article after or at the surface level of the main substrate. |
| Film Lamination | A distinct barrier film layer bonded or laminated onto the fiber substrate. |
| Surface Coating Systems | A surface-applied barrier layer formed from liquid, aqueous, bio-based, or similar coating materials. |
| Grease Resistance | An application-specific performance outcome influenced by food type, temperature, hold time, structure, and process consistency. |
Table 1. Core Terms Used in This Guide
3.1. PFAS-Free is a compliance-oriented claim, not a casual marketing phrase
In professional sourcing contexts, “PFAS-Free” should be understood as a claim that must be supported by a defined evidence chain. Depending on the market and certification framework, that evidence may include fluorine screening, targeted substance testing, signed declarations, ingredient documentation, and product-specific compliance review. It should never be treated as a stand-alone advertising phrase without method scope, document traceability, and sample identity.
3.2. Fluorine-free and PFAS-Free are related, but they are not identical concepts
“Fluorine-free” is often used in trade communication as shorthand for the absence of intentionally added fluorinated chemistry. However, fluorine-based screening and PFAS determination are not the same analytical question. A fluorine result may be used as an indicator or screening tool, while PFAS compliance may require a broader interpretation involving targeted analysis, total fluorine context, declarations, and technical documentation. In other words, a simple “non-detect” statement does not automatically mean that the product has been fully characterized under every relevant PFAS framework.
3.3. Internal additive systems are not the same as external barrier systems
In this article, Internal PFAS-Free Additive Systems refers to fluorine-free grease- and water-resistance chemistry introduced into the molded fiber process from within the pulp system. These additives are integrated into the production process before final forming and hot pressing. Their commercial value lies in process integration, scalable output, and reduced dependence on secondary surface treatment.
By contrast, External Barrier Systems refers to barrier structures added to the molded fiber article after or at the surface level of the main substrate. In this guide, external barrier systems include two distinct subcategories: Film Lamination and Surface Coating Systems. These routes may improve barrier performance in certain applications, but they should not be confused with internal additive technology.
3.4. Lamination and coating are not interchangeable terms
Lamination refers to a distinct barrier film layer that is bonded or laminated onto the fiber substrate. The technical logic is based on a separate material layer creating a physical barrier. Coating, by contrast, refers to a surface-applied barrier layer formed from liquid, aqueous, bio-based, or similar coating materials that are applied, dried, and fixed on the substrate surface. Both belong to external barrier systems, but they differ in structure, process flow, defect profile, and regulatory interpretation. For technical writing and sourcing review, they should never be treated as interchangeable language.
3.5. Grease resistance is an application-specific performance outcome
“Grease resistance” should not be read as a universal yes-or-no property. In molded fiber packaging, grease resistance depends on the actual food system, contact temperature, hold time, fill condition, structure design, wall thickness, and production consistency. A product that performs well for cold or ambient foods may fail under hot oil, hot sauces, or long delivery cycles. This is why the article later distinguishes between low-temperature internal additive systems and high-performance internal additive systems instead of treating all PFAS-Free additives as one category.
3.6. This article does not assume that one route is always correct
This guide does not assume that every external barrier solution is automatically inferior, nor that every internal additive system is automatically compliant or high-performing. The purpose of this white paper is narrower and more practical: to define the technical routes correctly, compare them under sourcing conditions, and help buyers decide which route is commercially, operationally, and regulatorily defensible for a given application.
4. Regulatory Baseline 2026 — What Buyers Must Actually Comply With
For buyers of PFAS-Free sugarcane tableware, regulatory review in 2026 cannot be reduced to a single certificate or a general supplier declaration. The compliance baseline is now shaped by a combination of market-specific PFAS thresholds, food-contact documentation requirements, and increasing scrutiny of barrier structures in fiber-based packaging. In practical sourcing terms, this means buyers must evaluate not only whether a product is promoted as PFAS-Free, but also how that claim is supported, which test logic was used, and whether the product structure remains commercially defensible under evolving packaging rules.
For export-facing procurement teams, the most important shift is that PFAS compliance is no longer a peripheral sustainability topic. It is becoming part of mainstream packaging approval, especially where food-contact safety, compostability claims, recyclability expectations, and plastic-reduction policies now intersect. In molded fiber foodservice packaging, this is particularly important because sugarcane tableware is often expected to meet several performance and compliance goals at the same time: grease resistance, heat tolerance, food-contact safety, and a credible circular-material profile.
4.1 European Union: PPWR has become the main 2026 reference point

For food-contact packaging placed on the EU market, the Packaging and Packaging Waste Regulation now provides the most important regulatory anchor for PFAS review. From August 12, 2026, food-contact packaging must not contain PFAS at or above the concentration limits set out in the regulation. These include a limit of 25 ppb for any individual PFAS, 250 ppb for the sum of PFAS, and 50 ppm for total PFAS. In addition, if total fluorine exceeds 50 mg/kg, manufacturers must be able to demonstrate the fluorine source as part of the technical documentation. For buyers, this means that a broad “PFAS-Free” claim is no longer enough. What matters is whether the supplier can provide a product-specific evidence package aligned with the applicable analytical logic and documentation burden.
The commercial implication is equally important. The EU packaging framework is not only tightening around substances of concern; it is also moving the market toward packaging structures that are easier to justify in a circular economy context. This is why fiber-based products with simpler material logic may offer a stronger long-term sourcing position than structures that depend heavily on additional barrier layers. For procurement teams, the question is no longer only whether a product can pass a test today, but whether its structure remains strategically defensible under the next phase of packaging scrutiny.
4.2 Why laminated or coated fiber structures require closer review in Europe
In the European regulatory context, buyers also need to distinguish carefully between molded fiber articles that remain primarily fiber-based and those that rely on plastic-containing barrier layers. EU guidance on single-use plastics has already made clear that paper or board products with plastic lining or plastic coating can still fall within a plastic-regulated interpretation. This does not mean that every laminated or coated structure is automatically non-compliant. It means that external barrier systems usually carry a higher interpretation burden, especially when buyers need to defend recyclability, repulpability, or plastic-reduction positioning in addition to PFAS compliance.
For this reason, laminated and coated molded fiber products should not be evaluated only on immediate barrier performance. Buyers should also consider whether the structure creates additional regulatory questions, recycling limitations, or market-access risks in regions where fiber purity and circularity are becoming more important purchasing criteria.
4.3 North America: certification logic, state restrictions, and FDA signals now work together
In North America, the compliance picture is shaped by three overlapping mechanisms: certification requirements, state-level food packaging restrictions, and federal regulatory action. For compostable foodservice products, BPI remains one of the clearest reference points. Its fluorinated chemicals policy requires evidence that the product formula does not contain fluorinated chemicals, test results from a BPI-approved lab showing a maximum of 100 ppm total organic fluorine, and a signed statement confirming that no intentionally added fluorinated chemicals are present. For sourcing teams, the significance of this requirement is not just the number itself. It is the fact that compliance must be supported by both documentation and laboratory evidence.
State-level regulation adds another layer of accountability. Washington State, for example, requires certificates of compliance for restricted food packaging categories and makes clear that manufacturers must retain these records while the packaging remains in use and for a defined period after distribution. This moves PFAS compliance beyond marketing language and into auditable supply-chain documentation. Buyers working with molded fiber bowls, plates, open-top containers, or closed containers should therefore expect not only claims, but also traceable compliance records that map to actual product types.
At the federal level, FDA action has further reinforced the market shift away from PFAS-based grease-proofing in paper and paperboard food packaging. The agency determined that 35 PFAS-related food-contact notifications are no longer effective as of January 6, 2025, with a compliance date of June 30, 2025 for certain food paper packaging produced, supplied, or used before that effective date. For procurement professionals, this is a strong signal that PFAS-free conversion is no longer a speculative trend. It is already embedded in the regulatory direction of the North American packaging market.
4.4 What this means for buyers of PFAS-Free sugarcane tableware
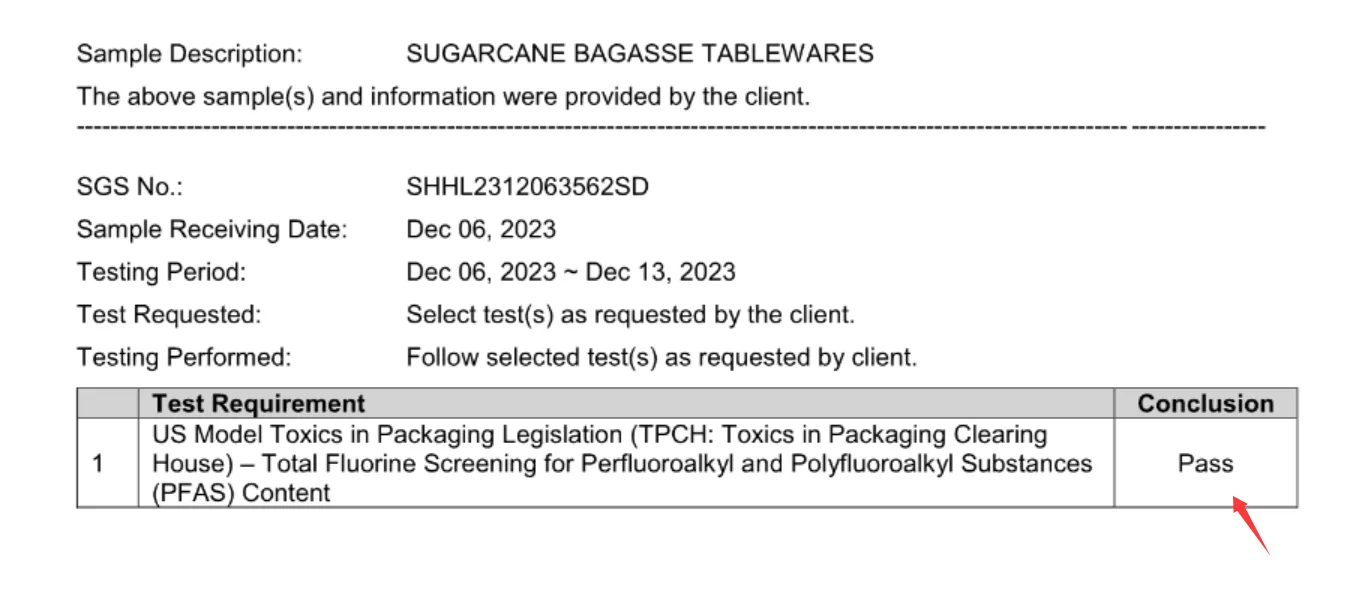 | 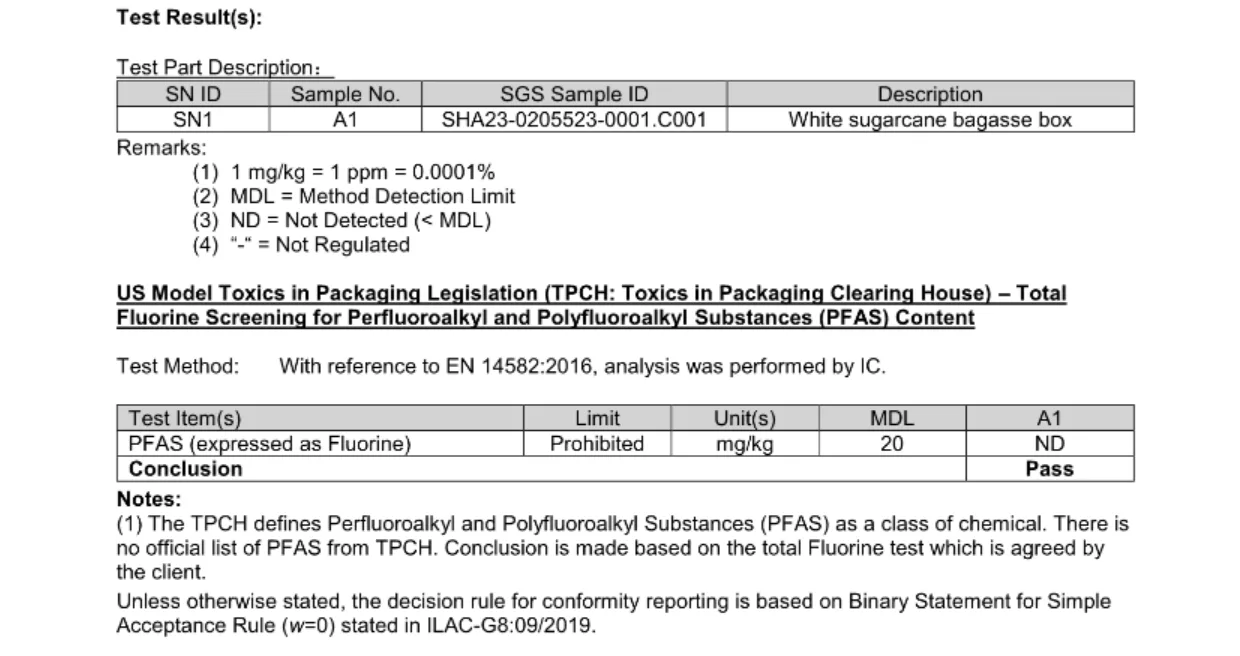 |
The procurement consequence is straightforward: buyers should no longer approve PFAS-Free bagasse tableware on the basis of a single logo, a generic declaration, or a one-time sample test. The 2026 baseline requires a more structured review. At minimum, buyers should confirm which market standard is being targeted, what type of fluorine or PFAS testing has been used, whether the test result matches the actual SKU and product construction, and whether the product relies on an internal additive system or an external barrier structure that may create additional regulatory or recycling questions.
In other words, regulatory compliance and technical design can no longer be reviewed separately. For PFAS-Free sugarcane tableware, the defensible sourcing decision is the one that aligns claim language, test scope, product structure, and market destination in a single approval logic. That is the foundation for the technical comparison that follows in the next sections.
5. Why Grease Resistance in Molded Fiber Is a Process Problem, Not a Slogan
In molded fiber foodservice packaging, grease resistance is often discussed as if it were a simple product feature. In reality, it is a process-controlled performance outcome. A sugarcane bowl, tray, or clamshell does not become commercially reliable just because it is labeled PFAS-Free, compostable, or molded from plant fiber. What determines real-world performance is whether the full production system can create a fiber structure that remains stable when exposed to oil, heat, moisture, pressure, and time.
This distinction is critical for buyers because molded fiber does not fail in the same way that plastic or film-based packaging fails. In sugarcane tableware, grease migration is usually linked to fiber porosity, local weak points, structural density, and processing consistency. A product may look visually acceptable at first filling, yet still develop backside oil shadowing, rim seepage, local softening, or bottom-panel penetration after several minutes of hot food contact. For this reason, grease resistance should never be evaluated only by appearance at the moment of use. It must be evaluated under defined application conditions.
5.1 Fiber structure is the starting point, not the final answer

Sugarcane bagasse is a strong and commercially attractive molded fiber substrate, but it is not naturally a high-barrier material. Like other plant-fiber structures, it contains a network of pores, fiber intersections, and variable-density regions created during forming, dewatering, and hot pressing. These structural features are exactly what make the product lightweight, compostable, and manufacturable at scale. They are also the reason uncontrolled oil and moisture penetration can occur if barrier design is weak or production conditions are unstable.
This is why a PFAS-Free molded fiber article should never be judged only by material identity. “Bagasse” is not a performance guarantee by itself. Two products made from the same base fiber can behave very differently depending on additive system, mold design, wall thickness, press conditions, dewatering efficiency, and post-forming consistency. In commercial sourcing, the real technical question is not whether the substrate is sustainable. It is whether the finished structure is engineered and produced to survive the intended food-contact load.
5.2 Grease failure is usually a structural event before it becomes a visible complaint
Many buyers assess grease resistance only after a visible leakage issue appears. That is too late. In practice, molded fiber failure often begins as a structural weakness long before it becomes an obvious customer complaint. Oil can move through lower-density regions, edge transitions, corner geometry, sealing rims, or stressed areas created during stacking and transport. By the time a product shows clear staining or leak-through, the underlying barrier system has often been underperforming for some time.
This is also why center-panel performance alone is not enough. A bowl bottom may appear stable in a static bench test, while the rim area, sidewall transition, or compartment edge becomes the real failure point during actual takeaway use. Buyers should therefore treat grease resistance as a full-structure performance issue, not a flat-surface laboratory claim.
5.3 Heat, oil load, and hold time are different variables and should not be mixed together
 |  |
One of the most common sourcing mistakes is to describe a food application too broadly. “Hot food” is not a test method. A molded fiber package filled with warm rice behaves very differently from one holding fried chicken, curry, oily noodles, or soup with a fat-rich surface layer. Temperature matters, but temperature alone does not explain performance. Oil load, contact duration, and product geometry are equally important.
This is why a PFAS-Free product that performs well for short contact with warm food may still fail under longer delivery cycles, higher grease concentration, or repeated heat retention. It is also why low-temperature internal additive systems and high-performance internal additive systems should not be grouped together as if they served the same application range. The difference between them is not just a marketing tier. It is a difference in intended use conditions and process capability.
| Variable | Why It Matters | Typical Buyer Mistake |
|---|---|---|
| Food Temperature | Higher temperature can accelerate fiber softening, moisture migration, and barrier stress. | Treating all “hot food” as one condition. |
| Oil Load | Grease-rich foods create a different penetration challenge from dry or low-fat foods. | Assuming warm rice and oily noodles require the same barrier level. |
| Hold Time | Longer contact duration increases the risk of backside staining, seepage, and structural weakening. | Approving a sample based only on short contact testing. |
| Product Geometry | Corners, rims, sidewall transitions, and compartment edges are often the first failure points. | Evaluating only the center panel or flat bottom area. |
| Wall Thickness | Thin-wall areas are more sensitive to porosity variation and grease breakthrough. | Reducing weight without revalidating barrier performance. |
| Process Consistency | Stable grease resistance depends on repeatable forming, dewatering, and hot-press conditions. | Judging performance from a single sample batch only. |
Table 2. Key Variables That Affect Grease Resistance in Molded Fiber Packaging
5.4 Condensation, moisture, and grease are not the same failure mode
Another common source of confusion is the misreading of moisture-related effects. In molded fiber packaging, condensation and grease penetration are not identical events. A hot-filled product may generate visible moisture or surface dampness due to steam migration and temperature difference, especially during lidded takeaway use. This does not automatically mean that the grease-resistance system has failed. The more important question is whether there is actual oil breakthrough, fiber softening, structural weakening, or persistent backside staining linked to fat migration.
For serious product evaluation, buyers should separate at least three conditions: hot moisture exposure, hot oil exposure, and combined heat-fat-hold-time stress. A supplier who cannot distinguish these conditions in testing or product recommendation is unlikely to provide reliable PFAS-Free guidance for real foodservice use.
5.5 Grease resistance in 2026 is a factory-discipline issue
By 2026, the market is no longer asking only whether a PFAS-Free molded fiber product can pass an isolated sample test. The more important question is whether the same performance can be maintained across commercial production, multiple SKUs, and repeat export batches. This is why grease resistance should be treated as a manufacturing discipline issue. Additive chemistry matters, but so do pulp preparation, dosage control, forming stability, mold temperature, dwell time, wall-thickness variation, and quality-control discipline.
From a sourcing perspective, this is the point where many suppliers begin to separate. Some can make an acceptable laboratory sample. Fewer can maintain repeatable performance when production speed increases, carton quantity scales up, and multiple product formats run in parallel. For buyers, the real purchasing risk is rarely the first sample. It is inconsistency at commercial scale.
This is also where Bioleader’s production experience becomes relevant. In molded fiber export supply, grease resistance is not managed as a single-material claim. It is managed as a combined result of fiber structure, process control, additive selection, and application matching. That is why the next section focuses first on internal PFAS-Free additive systems, including the practical difference between low-temperature and high-performance routes, before moving to external barrier systems such as lamination and surface coatings.
6. Route A — Internal PFAS-Free Additive Systems
Among the available PFAS-Free technology pathways for molded fiber foodservice packaging, internal additive systems remain the most commercially relevant starting point. Their importance comes from one practical fact: they are designed to build grease- and moisture-resistance performance within the molded fiber process itself, rather than relying primarily on a separate post-forming barrier layer. For buyers, this matters because integrated systems are generally easier to scale, easier to control in production, and easier to evaluate as part of a full factory process rather than as an isolated surface treatment.
In commercial sourcing, internal additive systems are often the route that best balances performance, throughput, and cost for mainstream sugarcane tableware applications. They are not automatically the strongest barrier option in every extreme use case, but they are often the most practical route for high-volume takeaway packaging where buyers need repeatable grease resistance without adding excessive structural complexity. This is also why internal systems should not be discussed as a single generic category. In real procurement and manufacturing, there is a major difference between low-temperature internal additive systems and high-performance internal additive systems.
 |  |
6.1 What internal additive systems actually mean
In this article, internal PFAS-Free additive systems refer to fluorine-free grease- and water-resistance chemistry introduced from within the molded fiber production process, typically through the pulp system before final forming and hot pressing. The commercial logic is straightforward: rather than building the barrier mainly through a separate external film or coating layer, the producer seeks to improve the final article’s resistance performance through process-integrated chemistry combined with forming, dewatering, and hot-press control.
This distinction is important because internal systems are often misunderstood as if they were merely “materials added to the pulp.” In practice, the additive itself is only one part of the result. Real performance depends on how the additive interacts with fiber distribution, structural density, moisture removal, and thermal forming conditions. This is why two suppliers may both claim to use PFAS-Free internal additives, yet deliver very different performance once the product is filled with hot, oily, or long-hold takeaway foods.
From a sourcing standpoint, the main strength of an internal system is that it can be assessed as part of a complete manufacturing route. Buyers are not only reviewing a chemistry claim. They are reviewing whether the supplier can maintain a stable process window across pulp preparation, additive control, forming consistency, hot pressing, SKU variation, and final batch output.
6.2 Low-temperature internal additive systems
Low-temperature internal additive systems are generally intended for less demanding grease-resistance environments. In practical terms, these systems are typically better suited to cold-chain, ambient, dry-food, or low-grease applications where the package is not exposed to prolonged hot oil, heavy fat loading, or long high-temperature holding conditions. They often remain commercially attractive because they are closer to standard production economics and can support mainstream volume manufacturing with a lower performance-cost burden.
This type of system can be fully appropriate when the application is correctly matched. For example, products used for salads, fruit, bakery items, room-temperature meals, or short-duration contact with relatively low-fat foods may not require the higher barrier margin of a more demanding internal system. In these cases, overengineering the product may only add cost without delivering real commercial value.
The sourcing risk begins when buyers or suppliers treat low-temperature systems as if they were universally suitable for all PFAS-Free molded fiber packaging. They are not. Once the food profile shifts toward hotter, oil-rich, sauce-heavy, or longer-hold takeaway use, the performance window changes significantly. A product approved for light-duty conditions may then show backside shadowing, edge seepage, local softening, or unstable consistency in real foodservice use. For this reason, a low-temperature internal system should be approved only against the actual application profile, not against a broad generic claim such as “hot food safe” or “grease resistant.”
6.3 High-performance internal additive systems
High-performance internal additive systems are developed for more demanding molded fiber applications where grease load, temperature stress, and hold time are materially higher. In sourcing terms, this is the route most relevant to hot takeaway meals, oily prepared foods, sauce-heavy applications, and more demanding foodservice environments. Their commercial value lies in offering stronger grease-resistance performance while still retaining the operational advantages of an integrated molded fiber process.
From a technical perspective, high-performance systems should not be understood as “the same additive used at a higher level.” In well-controlled production, they are better understood as a combination of internal additive chemistry, fiber-surface interaction, process stabilization, and hot-press densification. The final barrier effect is influenced by how effectively the system works across the full structure of the article, including lower-density regions, sidewall transitions, corners, rims, and other areas where molded fiber products usually fail first under real takeaway use.
This is also the point where process discipline becomes decisive. A high-performance internal system can only justify its claim if the factory can maintain stability across pulp consistency, additive dosing, mixing order, dewatering behavior, mold temperature, dwell time, and wall-thickness control. Without that discipline, even a technically advanced additive package may produce inconsistent commercial results. Based on Bioleader’s molded fiber production experience, the most reliable high-performance PFAS-Free outcomes are achieved not through a single-material claim, but through a repeatable process window that remains stable across different SKUs and export batches.
For buyers, the practical meaning is clear: high-performance internal systems are often the most balanced option for mainstream hot takeaway packaging, but they should still be validated against the actual use case. Heat level, grease concentration, fill time, lid condition, stacking pressure, and delivery duration all affect the final result. The right approval question is therefore not whether a supplier says the product is “high-performance,” but whether that performance has been demonstrated under conditions that reflect the intended application.
6.4 Why internal systems remain commercially attractive
The continuing strength of internal PFAS-Free additive systems is not only technical. It is also operational and commercial. Because the barrier logic is integrated into the molded fiber production route, buyers often gain a more scalable solution for mainstream foodservice packaging than they would through an external barrier structure that depends on separate downstream processing. This usually supports a cleaner production flow, fewer secondary-process variables, and a more practical cost structure for high-volume export supply.
Internal systems may also offer a more straightforward sourcing narrative in markets where buyers increasingly value simpler fiber-based packaging structures. They do not eliminate the need for testing, documentation, or application-specific validation, but they often reduce the structural complexity that comes with laminated or separately coated packaging. For procurement teams, that can mean a more manageable balance between barrier performance, production efficiency, and long-term compliance positioning.
6.5 Where internal systems still fail in practice
Despite their advantages, internal PFAS-Free additive systems do not remove technical risk. They can still fail when the application is misclassified, when the product has been over-lightweighted, when sharp geometry creates local stress points, or when process consistency is weak. In real-world foodservice packaging, common problem zones include sealing rims, compartment transitions, sharp corners, thin-wall regions, and formats exposed to long hold times with high-fat food systems.
This is why buyers should avoid treating internal systems as an automatic guarantee. A PFAS-Free internal additive route can be commercially strong, but only when the product design, process window, and application profile are aligned. In practical sourcing, this means the strongest suppliers are not the ones who make the broadest claims. They are the ones who can explain where the performance boundary lies, how it has been tested, and how repeatability is controlled at factory scale.
7. Route B — External Barrier Systems
External barrier systems represent the second major PFAS-Free pathway in molded fiber foodservice packaging. Unlike internal additive systems, which build resistance performance within the forming process itself, external barrier systems rely on a distinct surface-level barrier to improve grease or moisture protection. In sourcing terms, this route is usually considered when buyers need a stronger barrier margin than a standard molded fiber structure can provide through internal chemistry alone, or when a specific food application creates unusually demanding liquid, oil, or hold-time conditions.
For procurement teams, the key issue is not whether external barrier systems can work. In many cases, they can. The more important question is whether the added barrier performance justifies the resulting increase in structural complexity, process dependency, cost, and circularity review. This is why external systems should not be treated as a default upgrade. They should be evaluated as a distinct technical route with its own advantages, limitations, and regulatory implications.

7.1 What belongs to external barrier systems
In this guide, external barrier systems include two main categories: film lamination and surface coating systems. Both are surface-oriented solutions, but they are not interchangeable in technical or sourcing terms. Film lamination creates a barrier by bonding a distinct film layer onto the molded fiber substrate. Surface coating systems create a barrier by applying a liquid or dispersion-based material to the surface and forming a functional layer after drying, curing, or fixation.
This distinction matters because the two routes behave differently in production, in performance stability, and in market interpretation. A laminated structure may provide a clearer physical barrier concept, but it also introduces a more explicit multi-material logic. A coated structure may appear lighter or less visible in final appearance, but it can still add complexity through additional processing steps, coating uniformity control, and recyclability or repulpability questions. Buyers should therefore assess external systems not as a single category, but as two related yet distinct sourcing routes.
7.2 Film lamination

Film lamination is typically selected when a molded fiber article requires a more robust barrier layer for liquid, grease, or extended contact conditions. In commercial terms, the attraction of lamination is clear: it can create a stronger and more obvious barrier effect than many standard molded fiber solutions, particularly where the application involves difficult liquid-fat combinations or demanding holding conditions.
However, that higher barrier potential comes with trade-offs. A laminated article is no longer defined only by the molded fiber structure itself. It becomes a composite structure whose performance depends on the bonding quality, film integrity, edge stability, and compatibility between substrate and film layer. This introduces additional review points for buyers, not only in barrier performance, but also in downstream waste handling, circularity positioning, and market interpretation where simpler fiber structures may be favored.

From a sourcing perspective, film lamination should therefore be treated as a project-based solution rather than a universal improvement route. It can be commercially justified in niche, high-barrier applications, but it often weakens the simplicity advantage that makes molded fiber attractive in the first place.
7.3 Surface coating systems
Surface coating systems use a coated barrier layer rather than a bonded film. In practical manufacturing terms, this route introduces a separate surface-treatment step intended to improve grease or moisture resistance after the main molded fiber article has already been formed. Depending on the system, this may involve additional application control, drying, curing, or inspection steps before the final product is ready for packing and export.
The main advantage of coating systems is flexibility. In some applications, they can improve barrier performance without introducing the same type of visible film structure associated with lamination. In theory, this may allow buyers to target a stronger barrier outcome while retaining a more fiber-like visual or structural profile. But in practice, coating systems are rarely simple. Their performance depends heavily on coating distribution, layer consistency, edge coverage, defect control, and stability during commercial throughput.
This is also why spray-applied grease barriers are no longer the center of discussion in mainstream molded fiber sourcing. They may still appear in niche technical conversations, but for large-scale foodservice packaging, buyers usually care less about the application method itself and more about the repeatability, cost, and defect risk of the resulting coated structure.
7.4 Repulpability and circularity concerns
External barrier systems often face closer scrutiny because they can complicate the material story of molded fiber packaging. A simple molded fiber article already has a clear commercial identity: it is a fiber-based structure that can be positioned around compostability, reduced plastic content, or easier alignment with fiber-oriented circularity goals, depending on market and end-of-life route. Once a separate film or coating layer is added, that simplicity is reduced.
For buyers, this does not automatically mean the product becomes commercially unacceptable. It means that additional questions need to be asked. Can the structure still be justified under the buyer’s circularity claims? Does the barrier layer create downstream repulpability concerns? Does it add uncertainty in regions where plastic-related scrutiny remains high? These are not secondary marketing issues. They are now part of the sourcing decision itself.
This is one reason why many buyers continue to prefer a well-executed internal PFAS-Free system for mainstream takeaway applications whenever it can meet the real barrier requirement. From Bioleader’s perspective, external barrier systems should be reserved for cases where the application genuinely requires them, not simply as a default upgrade in the absence of proper process validation.
7.5 When external barrier systems are justified
External barrier systems are justified when the application demands a barrier performance level that cannot be delivered consistently by an internal molded fiber route under realistic commercial conditions. This may include special liquid packaging formats, unusually long hold-time conditions, food systems with extreme oil or sauce load, or other niche requirements where a stronger barrier layer is commercially necessary.
In such cases, buyers should still resist the temptation to treat external systems as a shortcut. The correct sourcing question is not whether the barrier looks stronger on paper, but whether the full structure remains commercially workable after accounting for production complexity, defect control, batch repeatability, and destination-market expectations. Used selectively, external barrier systems can solve real packaging problems. Used too broadly, they can undermine the structural and commercial advantages that made molded fiber attractive in the first place.
8. Decision Matrix — Which Route Fits Which Foodservice Scenario
Once buyers understand the difference between internal additive systems and external barrier systems, the next step is application matching. This is where many sourcing decisions either become more disciplined or more expensive than necessary. The correct route is rarely determined by a single claim such as “grease resistant” or “PFAS-Free.” It is determined by how well the selected route matches the actual food system, use temperature, hold time, barrier demand, and commercial operating model.
For mainstream molded fiber foodservice packaging, the most useful way to compare routes is through a decision matrix rather than a marketing hierarchy. A stronger barrier is not always the better sourcing decision if it adds unnecessary process cost, structural complexity, or circularity burden. Likewise, a lower-cost route is not the right choice if it cannot survive real takeaway conditions. The purpose of this section is therefore not to declare one route universally superior. It is to help buyers identify which route is commercially and technically defensible for each category of use.
| Comparison Factor | Internal PFAS-Free Additive Systems | External Barrier Systems |
|---|---|---|
| Barrier Logic | Performance built into the molded fiber process through internal chemistry and process control. | Performance enhanced through a separate surface-level barrier layer such as lamination or coating. |
| Best-Fit Use Case | Mainstream takeaway, hot meals, ambient foods, and large-volume foodservice packaging. | Special high-barrier applications, difficult liquid-fat combinations, and niche demanding formats. |
| Production Complexity | Integrated into the primary molded fiber route. | Higher due to additional barrier-layer processing and defect-control requirements. |
| Commercial Scalability | Generally stronger for mainstream high-volume production. | More dependent on secondary processing stability and project-specific economics. |
| Structural Simplicity | Usually more favorable for buyers seeking simpler fiber-based packaging logic. | Can introduce a more complex composite or surface-treated structure. |
| Main Buyer Risk | Misclassifying the application or overestimating barrier capability. | Accepting higher complexity and cost without a truly necessary barrier gain. |
| Approval Priority | Application matching, process repeatability, and realistic hot-food validation. | Barrier necessity, defect control, structural review, and end-market acceptance logic. |
Table 3. Master Comparison of Internal PFAS-Free Additive Systems and External Barrier Systems
8.1 Application-based route selection
For most buyers, the more practical question is not “Which route is better in theory?” but “Which route is right for this food application?” That decision should be based on use condition, not category labels alone. A salad bowl, a curry tray, a fried food clamshell, and a soup container may all be sold under the same packaging family, yet they place very different demands on the barrier system. The table below provides a practical starting point for route selection based on real foodservice scenarios.
| Foodservice Scenario | Typical Packaging Stress | Recommended Route | Buyer Focus |
|---|---|---|---|
| Cold salads, fruit, bakery, ambient foods | Low grease, low thermal stress, shorter contact duration | Low-temperature internal additive systems | Avoid unnecessary over-specification and cost inflation. |
| Standard hot takeaway meals | Moderate heat, moderate grease, routine delivery duration | High-performance internal additive systems | Validate repeatability under realistic fill and hold conditions. |
| Oily noodles, curry, fried foods, sauce-heavy meals | Higher grease load, more difficult stress on rims and transitions | High-performance internal additive systems, or external barrier systems if internal performance is insufficient | Test actual food profile, not only generic heat resistance claims. |
| Long-hold delivery with hot, fatty food systems | Extended contact time, thermal retention, stacking and transport stress | High-performance internal systems first; external barrier systems if clearly justified by testing | Focus on hold time, structural weak points, and commercial batch consistency. |
| Special liquid-rich or high-barrier niche formats | High liquid challenge, difficult barrier margin, project-specific demands | External barrier systems | Confirm barrier necessity and evaluate added complexity before approval. |
Table 4. Application-Based Decision Matrix for PFAS-Free Sugarcane Tableware
8.2 The most expensive mistake is selecting the wrong route for the wrong problem
In practical sourcing, the biggest cost driver is often not the unit price quoted at the beginning of the project. It is route mismatch. A buyer may overpay for an external barrier system that was never necessary for the actual food application. Or, more commonly, a buyer may approve a low-spec internal route for a foodservice use that requires a higher barrier margin, only to face complaints, retesting, delayed approval, or replacement cost later. In both cases, the technical mistake becomes a commercial cost.
This is why the most defensible decision matrix is the one built around real use conditions, not generic packaging categories. Buyers should first define the food profile, thermal load, grease load, hold time, lid condition, and destination-market compliance context. Only then should they decide whether a low-temperature internal system, a high-performance internal system, or an external barrier system is commercially justified. That sequence is what turns PFAS-Free sourcing from a reactive packaging choice into a controlled procurement strategy.
9. Procurement Verification SOPs
For PFAS-Free sugarcane tableware, technical comparison alone is not enough. Buyers also need a repeatable approval workflow that can separate compliant, scalable suppliers from those relying on broad claims, incomplete reports, or one-off sample success. In practice, the most effective procurement workflow is not the one with the most paperwork. It is the one that connects claim language, test scope, SKU identity, factory execution, and destination-market requirements into one review logic.
This is especially important in PFAS-Free molded fiber sourcing because barrier performance and regulatory defensibility are no longer independent topics. A product may perform well in a short bench test while still lacking the right fluorine screening logic, SKU-level traceability, or destination-market documentation. Conversely, a supplier may hold relevant reports but still fail at batch repeatability, edge-area performance, or application matching. Serious procurement therefore requires a structured verification sequence rather than a single approval event.
9.1 Compliance file review SOP
The first approval gate should always be document review. Before performance testing begins, the buyer should confirm whether the supplier’s PFAS-Free claim is supported by a product-specific compliance file rather than a generic marketing declaration. At minimum, that file should identify the actual SKU under review, the product structure, the target market, the test type used, the test date, and the issuing body or laboratory. If the supplier cannot map the report to the exact product family being quoted, the claim is not yet procurement-ready.
For compostable foodservice packaging sold into North America, this review should also check whether the supplier’s evidence logic aligns with recognized market expectations such as BPI’s fluorinated chemicals policy. That policy requires three core elements: formula review through safety data sheet documentation, a BPI-approved lab result showing a maximum of 100 ppm total organic fluorine, and a signed statement that no intentionally added fluorinated chemicals are present. Buyers do not need to ask every supplier for BPI certification itself, but they should understand the document logic that serious PFAS-Free review now requires.
| Verification Item | Why It Matters | Minimum Buyer Check |
|---|---|---|
| SKU identity | A valid report must correspond to the quoted product, not a similar item. | Confirm item code, size, structure, and material match the offer. |
| PFAS / fluorine test scope | Different tests answer different questions. | Check whether the result is screening-only, targeted, or linked to a certification rule. |
| Date and batch traceability | Old or untraceable results weaken procurement defensibility. | Require recent reports and identifiable sample information. |
| Signed declaration | A lab report alone may not confirm intentional-use status. | Request a signed no intentionally added fluorinated chemicals statement where relevant. |
| Destination-market alignment | A report may be technically valid but commercially insufficient for the target market. | Check EU, North American, or customer-specific requirements before approval. |
Table 5. Minimum Compliance File Review for PFAS-Free Sugarcane Tableware
9.2 Fluorine screening SOP
The second approval gate is analytical screening. Buyers should treat fluorine screening as a necessary but limited tool. It can be highly useful for filtering risk, but it is not a complete substitute for broader regulatory interpretation, supplier declarations, or application-specific review. In professional sourcing, screening should answer a practical question: does this product show a fluorine profile that is compatible with the PFAS-Free claim being made?
For this reason, screening results should always be read together with the product description, barrier structure, and declaration package. A buyer should not assume that a simple “non-detect” statement automatically resolves every PFAS-related question, just as a low fluorine result does not automatically guarantee universal market acceptance. What matters is whether the test logic matches the claim logic. This is why Bioleader’s view is that fluorine screening should be used as an approval filter, not as the only approval basis.
9.3 Hot oil / hot soup performance SOP
The third approval gate is application testing. For molded fiber foodservice packaging, this is where many sourcing projects either become reliable or fail later in the market. The test should not be based on abstract statements such as “microwave safe” or “grease resistant.” It should be built around the actual use condition: fill type, food temperature, grease load, hold time, lid condition, and expected delivery or service duration.
A practical buyer SOP should therefore define at least five variables before testing begins: the medium used, the fill temperature, the hold time, the pass/fail observation points, and the number of samples. For example, a hot soup test and a hot oil test should not be treated as interchangeable, because moisture stress and grease stress are different mechanisms. Likewise, edge areas, base transitions, and rim zones should be inspected separately from the flat center panel, since these are common weak points in molded fiber structures.
| Test Element | Buyer Should Define | What to Observe |
|---|---|---|
| Test medium | Soup, oil-rich food, curry, noodles, or other application-relevant medium | Whether the package is stressed by moisture, grease, or both |
| Fill temperature | Realistic service or takeaway temperature range | Softening, staining, deformation, or early barrier loss |
| Hold time | Short service use, standard delivery time, or long hold condition | Delayed seepage, backside shadowing, edge failure |
| Inspection zones | Bottom panel, sidewall transition, corner, rim, compartment edge | Localized weak points rather than center-panel-only results |
| Sample size | A repeatable sample count, not a single-piece demonstration | Variation between pieces and consistency of failure mode |
Table 6. Practical Buyer SOP for Hot Oil / Hot Soup Validation
9.4 Factory execution audit SOP
The fourth approval gate is factory execution review. This is the step that most often separates technically credible suppliers from those that only perform well at sample stage. A buyer may receive an acceptable PFAS-Free sample, but the commercial risk does not begin there. It begins when the product moves into normal production speed, multi-SKU scheduling, export packing, and shipment-level quantity.
For this reason, factory review should focus on repeatability rather than only equipment description. Buyers should ask whether the supplier can explain how additive dosage is controlled, how batch variation is managed, how wall-thickness consistency is monitored, and how failure zones are identified when performance drifts. In export practice, these questions are often more important than a polished product brochure. Based on Bioleader’s manufacturing and export experience, the strongest PFAS-Free suppliers are usually the ones that can describe their control logic clearly before problems occur, not after complaints appear.
Where destination-market rules require retained records, buyers should also check that the supplier can support document continuity. In Washington State, for example, manufacturers of restricted food packaging must keep a certificate of compliance on file while the packaging is in use and for three years after its last sale or distribution. That is exactly the type of documentation discipline buyers should now expect in serious PFAS-Free sourcing.
 |  |
10. Real-World Pitfalls Buyers Commonly Miss
Even when buyers understand the difference between internal additive systems and external barrier systems, sourcing mistakes still happen. In most cases, the problem is not a lack of technical terms. It is the tendency to approve packaging on the basis of simplified assumptions. PFAS-Free molded fiber packaging often looks straightforward on paper, but real commercial failure usually happens when one variable is treated as if it explains the whole product. A report is mistaken for a full approval. A sample is mistaken for scalable production. A clean appearance is mistaken for real barrier stability.
The following pitfalls appear repeatedly in molded fiber foodservice sourcing. They are not minor operational details. They are the reasons why projects that look compliant at quotation stage can still fail after launch, during export scaling, or in actual takeaway use.
10.1 PFAS-Free does not mean hot-oil ready
One of the most common errors in procurement is assuming that a PFAS-Free claim automatically implies strong hot-oil resistance. It does not. PFAS-Free only addresses one aspect of the product’s chemical positioning. It does not by itself define the barrier strength, thermal margin, or hold-time suitability of the molded fiber structure. A product may be PFAS-Free and still be appropriate only for low-grease or ambient applications.
This is why route selection matters. Buyers should approve a product based on its tested use condition, not on the existence of a PFAS-Free statement alone. In practice, the most reliable projects are the ones where the claim and the application are validated together from the start.
10.2 Sample success is not factory success
Another common mistake is over-relying on the first approved sample. A sample may be made under slower or more controlled conditions than a normal export batch. It may also represent only one mold condition, one operator setting, or one part of the process window. Once the project moves into full production, performance can shift if wall thickness varies, additive dosage drifts, or forming and hot-press conditions become less stable.
This is why commercial sourcing should never end at sample approval. Buyers should also ask how repeatability is controlled across volume production, multiple SKUs, and shipment-level output. In Bioleader’s experience, many preventable complaints do not start with wrong material selection alone. They start when buyers assume a good first sample automatically proves stable long-run execution.
10.3 Condensation is not always grease failure
Moisture on the outer surface of a molded fiber article is often misread as proof that the grease-resistance system has failed. In reality, hot filled packaging can generate condensation, dampness, or temporary surface darkening because steam and temperature difference create moisture movement through or around the fiber structure. That is not always the same as oil breakthrough.
The correct question is whether the package shows actual grease penetration, structural weakening, persistent backside staining, or barrier collapse under use conditions. Buyers who fail to distinguish condensation from grease migration may reject acceptable packaging for the wrong reason—or worse, approve unacceptable packaging because they inspected only moisture-related effects and not fat-related failure zones.
10.4 Edge and rim failures matter more than center-panel performance
Flat-surface testing can create false confidence. Many molded fiber products look strong in the center panel while failing first at the rim, sidewall transition, fold area, corner radius, or compartment edge. These are the places where fiber density can change, mechanical stress can concentrate, and filling or stacking loads can expose structural weakness.
For this reason, the most useful performance review is always structure-wide. Buyers should inspect the full article, not just the easiest surface to observe. A product that survives a center-panel check but fails around the rim is not a reliable takeaway solution.
10.5 Lightweighting can quietly destroy barrier stability
Cost pressure often pushes suppliers and buyers toward lightweighting. In some cases, that optimization is valid and commercially efficient. But if weight reduction is introduced without revalidating the barrier system, it can become one of the fastest ways to weaken PFAS-Free performance. Thin-wall areas are more vulnerable to porosity shifts, thermal distortion, and local breakthrough under grease or hold-time stress.
Lightweighting decisions should therefore never be treated as purely commercial changes. They are performance changes. The correct question is not whether the part still looks similar after weight reduction, but whether the updated structure still passes the same barrier and consistency requirements under realistic foodservice conditions.
| Common Pitfall | What Buyers Often Assume | What Should Be Checked Instead |
|---|---|---|
| PFAS-Free claim | The product is automatically suitable for hot greasy food. | Match the claim to a tested application profile. |
| Approved sample | Commercial production will perform the same way. | Audit repeatability and batch-control discipline. |
| Outer moisture | Any dampness means grease failure. | Separate condensation from actual grease breakthrough. |
| Center-panel pass | The full structure is safe. | Inspect rim, corner, transition, and edge zones. |
| Weight reduction | Lower weight means only lower cost. | Revalidate barrier stability after lightweighting. |
Table 7. Common Procurement Mistakes in PFAS-Free Molded Fiber Sourcing
In short, the most dangerous sourcing errors are usually not dramatic. They are small misreadings repeated through the approval process: treating a declaration as a full evidence package, treating a sample as proof of factory control, or treating one visible symptom as the whole failure mechanism. Buyers who avoid these mistakes usually do so not by being more skeptical in general, but by being more precise in how they define, test, and approve PFAS-Free packaging.
11. Exclusive Data Pack Framework
For PFAS-Free sugarcane tableware, strong writing alone does not create authority. In 2026, authority increasingly comes from evidence architecture: the ability to show what was tested, how it was tested, what failed, what passed, and under which production conditions the result remains repeatable. This is where many articles lose credibility. They describe performance in broad terms, but do not distinguish between laboratory observation, certification logic, factory data, and commercial inference.
For white-paper-level sourcing content, buyers should expect a clearer data structure. The most credible technical content does not simply say that a product is PFAS-Free, grease resistant, or suitable for takeaway. It identifies the evidence category behind each claim. That distinction is important not only for procurement review, but also for Google E-E-A-T and AI extraction logic, because content supported by clearly framed evidence is easier to trust, cite, and compare.
11.1 What data should be published
Published data should be the kind that a serious buyer can interpret without guessing. That usually includes the product identity, the test condition, the observation window, the pass or fail logic, and the boundary of the result. For example, a useful data point is not “excellent grease resistance.” A useful data point is a defined result observed under a stated condition, such as hot-oil exposure, hold time, or application-specific stress. The more specific the method logic, the more useful the published claim becomes.
For public-facing technical content, the best data points are the ones that help buyers compare routes without overstating universality. These may include barrier performance by food category, structure-related weak points, typical failure zones, repeatability observations across batches, and process-sensitive variables such as wall thickness or hold time sensitivity. Published correctly, such data supports not only sourcing decisions, but also the credibility of the article itself.
11.2 What data must be identified as internal observation
Some of the most valuable information in molded fiber sourcing comes from internal production and export experience rather than public certification databases. That can include complaint-rate patterns, batch-level variation, defect mapping, response to lightweighting, mold-related weak-point behavior, or observations from repeated foodservice application testing. This kind of information can be extremely useful, but it should always be presented honestly as internal observation, production experience, or factory-side validation rather than as universal industry fact.
That distinction is particularly important for Bioleader-style authority content. Internal experience is a strength, not a weakness, when it is framed correctly. Buyers value it because it reflects what happens at commercial scale, not only in certification language. But if internal observations are presented as though they were third-party universal rules, the article becomes easier to challenge. The stronger approach is to name the evidence category clearly and let the reader understand what type of conclusion is being made.
11.3 What claims should never be made without evidence
Some statements are too risky to make unless the evidence is explicit and product-specific. These include absolute phrases such as “100% leak-proof,” “fully PFAS-free” without method scope, “guaranteed 95°C performance” without condition definition, or “recyclable” and “repulpable” without a clear technical basis. These claims may look attractive in sales communication, but in procurement review they often create more risk than value.
The most defensible white paper is therefore not the one with the most aggressive claims. It is the one that makes the cleanest separation between certification-backed evidence, internal validation, observed process behavior, and project-specific inference. In Bioleader’s case, that means building authority not by exaggerating molded fiber performance, but by documenting where the route works, where it fails, and what buyers should verify before approval.
| Evidence Type | Suitable Use in This Article | How It Should Be Labeled |
|---|---|---|
| Official regulation / certification rule | Compliance baseline, threshold, legal timing, buyer requirements | Regulatory requirement / certification rule |
| Third-party test report | Product-specific analytical support or screening logic | Third-party report / laboratory result |
| Internal lab simulation | Application testing, hold-time comparison, structural weak-point analysis | Internal validation / internal test observation |
| Production observation | Batch repeatability, defect pattern, process-window commentary | Production experience / factory-side observation |
| Commercial inference | Cost-logic commentary, sourcing recommendation, decision framework | Sourcing judgment / commercial interpretation |
Table 8. Evidence Hierarchy for PFAS-Free Sugarcane Tableware Content and Sourcing Claims
12. Final Recommendations for 2026 Buyers
For buyers evaluating PFAS-Free sugarcane tableware in 2026, the most important conclusion is simple: do not purchase the claim before you validate the route. PFAS-Free is now a baseline expectation in many markets, but the correct sourcing decision still depends on how the barrier performance is delivered, how the structure behaves in real foodservice use, and how the product is documented for the destination market. The strongest procurement strategy is not the one that selects the most aggressive technical claim. It is the one that aligns regulation, application profile, and factory execution into one verifiable approval process.
For most mainstream takeaway and foodservice applications, high-performance internal PFAS-Free additive systems remain the most balanced route. They often provide the right combination of grease resistance, production efficiency, and manageable structural complexity. Low-temperature internal additive systems still have an important place in cold-chain, ambient, and lower-grease packaging, but only when the application is correctly matched. External barrier systems remain commercially valid for special high-barrier requirements, yet they should be selected for defined reasons, not as a default assumption that “more barrier must be better.”
Based on the comparison in this guide, buyers should follow five practical priorities. First, start from destination-market compliance, not from generic sustainability language. Second, separate fluorine screening from performance validation, because they answer different sourcing questions. Third, test the real use condition rather than relying on abstract statements such as hot-food safe or grease resistant. Fourth, review repeatability at factory scale rather than approving on sample appearance alone. Fifth, document the evidence chain clearly so that performance claims, compliance logic, and product structure can be defended together. This is the sourcing discipline that will matter most as PFAS-Free packaging moves deeper into regulated mainstream procurement.
From Bioleader’s perspective, the future of PFAS-Free molded fiber packaging will belong to suppliers that can combine process-controlled internal barrier performance, disciplined documentation, and application-specific validation. In other words, the market is moving away from packaging selected by label language and toward packaging selected by technical proof. Buyers who adapt to that shift now will reduce both compliance risk and commercial failure risk over the next phase of global packaging regulation.
13. FAQ
13.1 What reports are needed for PFAS-free food-contact packaging in the EU after August 12, 2026?
Buyers should expect a product-specific evidence package rather than a generic PFAS-Free statement. At minimum, the supplier should be able to explain the analytical basis of the PFAS claim, provide test documentation linked to the quoted SKU, and show technical documentation consistent with the EU packaging rule structure. If fluorine is present above the applicable threshold logic, the supplier should also be able to explain the fluorine source within the technical documentation framework. For procurement, the key issue is not just the report itself, but whether the report matches the actual product construction and market destination.
13.2 Does BPI certification automatically prove PFAS compliance?
No. BPI’s fluorinated chemicals policy provides a strong compliance logic for relevant compostable products, including formula review, laboratory testing, and a no intentionally added fluorinated chemicals declaration. However, buyers should not treat any one certification as a universal substitute for full market review. A certification result must still be read together with SKU identity, destination market, product structure, and the supplier’s current documentation status.
13.3 Does “non-detect” for total fluorine mean zero PFAS?
Not automatically. A non-detect result can be useful, but it depends on the analytical method, detection scope, and reporting framework. In procurement terms, “non-detect” should be treated as one element of the evidence package, not as a complete stand-alone conclusion. Buyers should still review what was tested, what was not tested, and how the result connects to the PFAS-Free claim being made.
13.4 Why can film lamination still trigger plastic-lining concerns in some markets?
Because film lamination changes the packaging from a simple fiber structure into a more complex surface-barrier or composite logic. In some regulatory and market contexts, paper or fiber products with plastic lining or plastic coating receive closer review under plastic-related rules, circularity discussions, or recycling expectations. The issue is not only whether the product performs well, but also whether the structure remains easy to justify under the buyer’s end-market packaging strategy.
13.5 Can coated molded fiber still be recyclable or repulpable, and how should buyers verify that?
Potentially yes, but buyers should not assume it by default. The correct question is whether the specific coated structure remains compatible with the intended recycling or repulping route and whether the supplier can support that position with technical reasoning or testing. Procurement teams should ask what kind of coating is used, how it affects the fiber structure, whether it changes downstream handling, and whether the supplier can explain the claim in a way that fits the destination market’s expectations.
Next Step for Buyers
For Enterprise Buyers
Need to review PFAS-Free molded fiber packaging for EU or North American markets? Bioleader can help you assess structure logic, documentation gaps, and application-specific barrier risks before commercial approval.
For Growing Food Brands
Starting a PFAS-Free packaging transition? Request Bioleader’s practical validation checklist to compare internal additive systems, external barrier systems, and real application risks more efficiently.
Bioleader supports buyers with molded fiber application review, PFAS-Free documentation coordination, and export-oriented packaging validation.
Copyright Notice:
© 2026 Bioleader®. If you wish to reproduce or reference this content, you must provide the original link and credit the source. Any unauthorized copying will be considered an infringement.



