
Introduction: The Hidden Cost of Non-Compliance
In 2026, the landscape for compostable packaging is shifting drastically. No longer a mere trend, compliance with global packaging regulations has become a legal imperative. For businesses still relying on outdated packaging materials, the consequences can be severe—both financially and legally.
Example: Imagine a $200,000 shipment of sugarcane bagasse tableware flagged by California Customs under AB 1200 due to PFAS contamination. The result? A complete denial of entry, storage fees, and the destruction of goods, followed by lawsuits for the lost revenue. This is not just a setback—it’s a liability risk that many businesses fail to anticipate.
In 2026, compliance is no longer just a logistical hurdle but a legal liability transfer. The importer of record (the buyer) bears 100% of the financial and legal burden if a supplier provides falsified or incomplete data. This makes the verification process a fiduciary duty for sourcing officers.
For small and medium-sized businesses, certifications like PFAS-Free are not just for compliance—they are a survival ticket. Without the correct certifications, even a small shipment can halt operations and waste capital. This guide will walk you through the critical certifications you need for compostable packaging, including BPI, OK Compost, and PFAS-Free certifications, while providing actionable insights into the audit process that helps safeguard your supply chain.
* Risk analysis based on 2026 California AB 1200 and New York S.8817 regulatory enforcement protocols.
I. North American Standards: The BPI & PFAS-Free Frontier
The North American market is a critical hub for the global packaging industry. However, new regulations are evolving rapidly, and businesses need to adapt quickly to avoid severe penalties or market rejection.
1. BPI Certification (ASTM D6400/D6868)
BPI Certification is a critical benchmark for industrial compostability in North America. Under ASTM D6400 and D6868, packaging materials must meet specific requirements to be certified compostable in industrial facilities.
Key Takeaway for Decision Makers:
A BPI certification confirms that the packaging breaks down in an industrial composting facility. Without this certification, your product could face rejection at customs and be subject to fines.
2. Critical Compliance Audit
One common mistake in BPI verification is overlooking the Maximum Thickness Limit. Under ASTM D6400, each material is certified only up to the tested thickness (e.g., 0.8mm for fiber plates). If you source a “heavy-duty” version (e.g., 1.2mm), the existing certificate becomes void. Bioleader offers a detailed Thickness-to-SKU Mapping Report, ensuring all your SKUs are within the certified physical boundaries.
Key Takeaway for Decision Makers:
Failure to match thickness specifications could invalidate your certification. Ensure your procurement team performs a Physical-Spec Matching Audit before purchasing.
3. The PFAS-Free Mandate
PFAS (Per- and Polyfluoroalkyl Substances) have been used in packaging to provide water- and grease-resistance, but due to their toxicity and environmental persistence, they are being phased out in North America. For 2026, PFAS-free certification is a legal requirement in many states, including California and New York.
Why Total Fluorine (TF) is No Longer Sufficient:
TF measures all fluorine atoms, including naturally occurring minerals. However, TOF (Total Organic Fluorine) focuses on synthetic PFAS chemicals. Customs authorities now require TOF testing with a maximum threshold of 100ppm.
Key Takeaway for Decision Makers:
Don’t accept “Fluorine-Free” statements. Only suppliers who provide TOF testing reports (using Combustion Ion Chromatography (CIC)) with TOF levels below 100ppm can ensure your packaging is compliant with the 2026 PFAS ban regulations.
Certification Verification Roadmap
A technical timeline for ensuring 100% regulatory compliance in 2026.
| Step | Key Action | Lead Time |
|---|---|---|
| 01 | Raw Material Screening: Rapid TF screening to ensure initial purity, identifying potential contaminants. | Week 1 |
| 02 | TOF Quantitative Analysis: Utilizing the CIC Method (<100ppm) to verify total organic fluorine content for PFAS compliance. | Weeks 2-4 |
| 03 | Master Certificate Issuance: Final BPI/TÜV verification to confirm compliance with industry standards. | Week 8 |
| 04 | Strategic Sub-license: Transfer of usage rights for private labels, enabling brand compliance without full certification costs. | Week 10 |
Technical Forensic
Combustion Ion Chromatography (CIC) offers a definitive “fingerprint” of synthetic PFAS chains, ensuring legal compliance. Bioleader adheres to an internal standard with TOF < 50ppm, which exceeds industry thresholds and provides an additional “buffer zone” during customs inspections.
4. FDA Food Contact Safety
Ensuring that food-contact packaging meets FDA food safety standards is essential for businesses operating in North America. These standards ensure that food packaging materials do not leach harmful chemicals into food.
Bioleader’s Commitment:
All Bioleader materials, including Bagasse and Cornstarch-based products, undergo rigorous testing according to the FDA Food Contact Substances (FCS) guidelines to ensure zero toxic migration and confirm they are safe for direct food contact.
Key Takeaway for Decision Makers:
FDA certification ensures that your materials comply with U.S. food safety laws and do not leach harmful chemicals into food, making your products safe for consumers.
5. Audit Dimension Checklist
This checklist serves as a practical tool for sourcing officers and procurement managers to verify the compliance of suppliers. It helps ensure the materials you source are fully compliant with regulatory standards, minimizing the risks associated with customs rejection and legal liabilities. Below is a summary of key compliance benchmarks to guide your procurement decisions.
| Audit Dimension | Compliance Benchmark | Risk Warning |
|---|---|---|
| Certificate Consistency | The certificate holder must match the Shipper listed on the Bill of Lading (BL). | Using a related factory certificate without authorization may be deemed fraudulent. |
| PFAS Testing Method | Must be labeled as TOF (CIC Method), with a threshold of <100ppm. | A “Fluorine-Free” statement without scientific backing has no legal defense. |
| Specification Matching | The product’s actual thickness must not exceed the certified maximum thickness (e.g., 0.8mm). | Exceeding 0.05mm in thickness could invalidate compliance. |
| Laboratory Accreditation | Must have ISO/IEC 17025 certification. | Data from non-accredited laboratories is not accepted by customs. |
| Note: This table is an essential tool for verifying the compliance of your packaging materials. Ensure your supplier’s documentation is fully accurate to avoid compliance risks. Looking to verify our credentials? [View the Bioleader Official Certification & Lab Report Library] — Explore our full range of BPI, OK Compost, and TOF reports to ensure your 2026 shipment is 100% compliant. | ||
Summary & Key Takeaways
- BPI Certification is essential for compostable packaging in North America and ensures market acceptance in industrial composting facilities.
- PFAS-Free Certification is a legal requirement in many U.S. states, and TOF testing is crucial for compliance.
- FDA Food Contact Safety Certification ensures that food packaging materials are safe for food contact and comply with U.S. food safety regulations.
II. European Standards: OK Compost & The SUPD Filter
Europe remains one of the most demanding markets for compostable food packaging, not because of one single certificate, but because of the way standards, labels, and product-specific marking rules interact. For sourcing managers, the European challenge is not simply “Is this material compostable?” but rather, “Is this exact SKU certified for the right end-of-life pathway, and is it marketed with the correct legal message?” This technical analysis outlines the key European filters that determine whether a product can move smoothly into distribution channels or become a compliance liability.
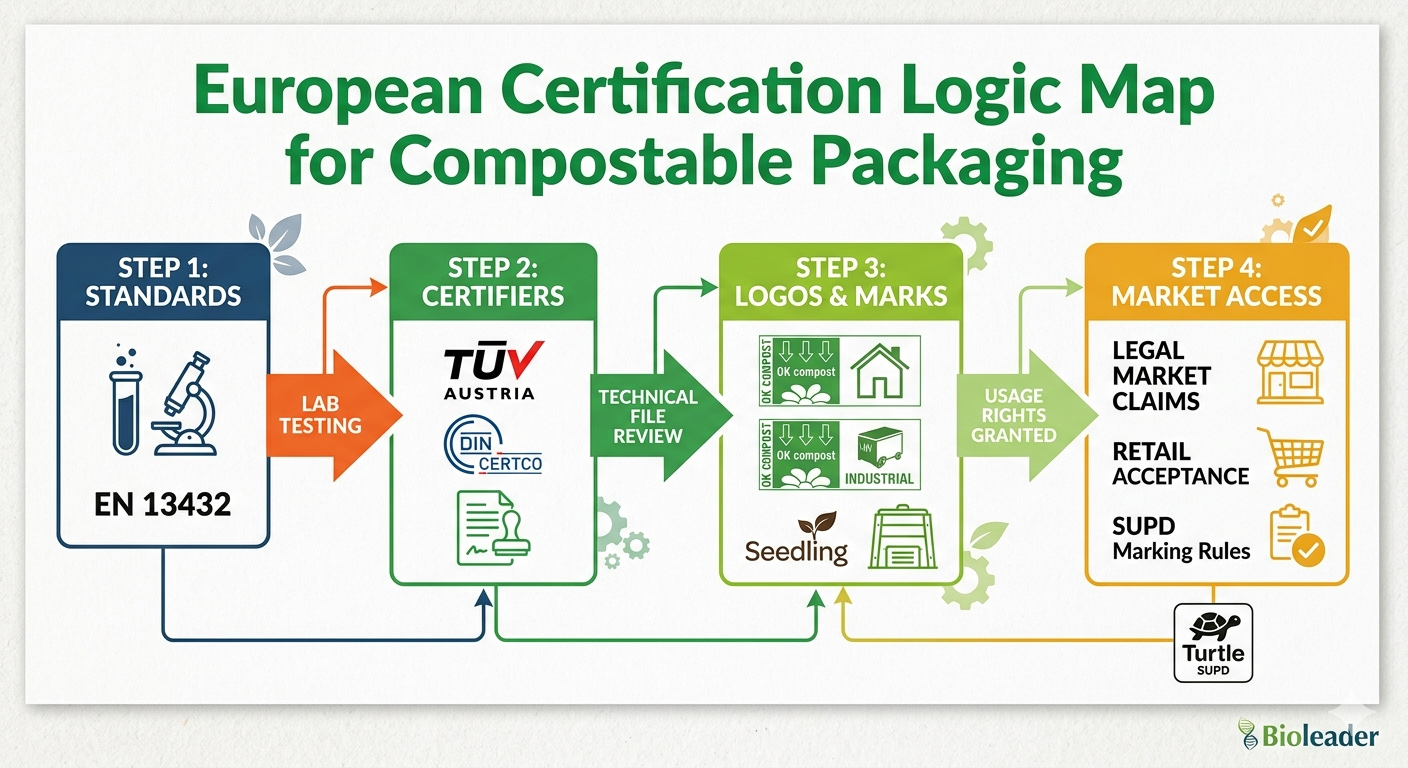
1. EN 13432: The Legal Foundation of Industrial Compostability in Europe
The European standard EN 13432 is the core reference point for packaging claimed as industrially compostable. In practical sourcing terms, it is the benchmark used to evaluate whether packaging can disintegrate and biodegrade under controlled industrial composting conditions without causing unacceptable environmental residues. It is also the standard underlying the Seedling certification scheme used across Europe for industrially compostable products.
 |  |
For buyers, the critical point is this: EN 13432 is not a marketing slogan. It is a technical compliance framework. If a supplier claims “eco-friendly,” “biodegradable,” or “green,” but cannot map the claim back to an EN 13432-based certification route, the product may have weak legal defensibility in the EU market. This is especially important for private-label importers, because under customs and retailer review, the burden of proof sits with the economic operator placing the packaging on the market. This is why professional sourcing must move beyond generic environmental language and require certificate-backed conformity.
2. TÜV AUSTRIA OK Compost HOME vs. OK Compost INDUSTRIAL
One of the most misunderstood points in compostable packaging is the difference between home compostability and industrial compostability. TÜV AUSTRIA’s certification systems make this difference visible and commercially actionable.
OK compost INDUSTRIAL confirms that a product is suitable for controlled composting facilities, where temperature, humidity, oxygen, and processing time are managed. This is the pathway behind most large-scale compostable packaging programs in Europe. The Seedling mark is also tied to this industrial compostability route.
 |  |
OK compost HOME, by contrast, is materially stricter in commercial terms because it signals that a product can biodegrade under lower-temperature, less controlled backyard composting conditions. That is why this label carries strong premium value for brands selling into retail, specialty organic stores, and sustainability-driven foodservice chains. It is not simply another logo; it is a higher consumer-facing trust signal.

For procurement teams, the practical implication is straightforward:
If your target buyer is a municipality, waste program, or institutional buyer, industrial compostability may be sufficient.
If your target buyer is a premium retailer or a consumer-facing private label that wants a stronger sustainability story, home compostability can become a decisive commercial differentiator.
This is where many sourcing errors occur. A supplier may provide a valid industrial compostability certificate, but the buyer may market the product as “home compostable” without legal support. That mismatch can create retailer disputes, consumer complaints, or forced relabelling. Professional sourcing, therefore, requires that the end-of-life claim on pack exactly match the certificate scope.
3. Seedling Mark: What It Really Signals
The Seedling logo is not a decorative eco-symbol. It is a registered trademark owned by European Bioplastics and is used only for products formally certified as industrially compostable according to EN 13432 through approved certification bodies such as DIN CERTCO and TÜV AUSTRIA Belgium.
 |  |
This matters because many buyers still confuse a test report with a certification logo right. A positive laboratory result alone does not grant the right to use the Seedling symbol. The use of the Seedling mark is strictly regulated under the European Bioplastics Labeling Guidelines. The right to display the mark depends on the official certification pathway and the specific product scope. For large distributors, this distinction affects legal labelling. For small private-label brands, it affects whether the packaging can be marketed as certified compostable without trademark exposure.
In commercial terms, the Seedling logo functions as a market access shortcut. It reduces the burden of explanation in front of buyers, retailers, and auditors because it translates technical compliance into a visual trust signal. But only when the logo use is legitimately backed by the relevant certification file.
4. SUPD: The Plastic Content Filter and Marking Risk
The Single-Use Plastics Directive (SUPD) adds another layer of complexity because it is not limited to compostability. It also introduces marking requirements for certain single-use plastic products and product categories placed on the EU market. The European Commission provides harmonised marking specifications and makes clear that required labels must follow official rules for content, size, colour, and placement.
For sourcing managers, this means a product can be “green” in one sense and still trigger plastic-related marketing obligations in another, depending on its composition and how the law classifies it. This is especially relevant for coated paper products, cups, lids, and composite structures.
⚠️ 2026 Regulatory Alert: The ‘Hidden Plastic’ Trap
In 2026, many EU member states have increased randomized lab testing for ‘hidden plastics’ (polymers used in binders or coatings). Even if a product is EN 13432 certified, if it contains traces of non-exempted polymers, it must carry the mandatory SUPD ‘Plastic in Product’ marking (the “Turtle” logo).
Bioleader’s compliance team helps clients navigate this ‘Grey Zone’ to determine whether your final SKU needs compostability certification, SUPD marking, or a combination of the three, avoiding severe mislabeling penalties.
III. Material-Specific Certification Matrix
The matrix below is designed as a procurement decision tool, not a generic marketing chart. It helps both large buyers and emerging brands understand what certification combination is typically expected for different material systems. Before selecting a certification route, buyers should first understand the structural and end-of-life differences between material families.
Important technical note:
The Thickness Limit shown below should be treated as a typical sourcing checkpoint, not a universal legal number. Final compliance always depends on the specific certificate appendix, the tested formulation, and the exact SKU geometry approved by the certifier.
1. Material-Specific Certification Matrix
| Material | BPI (NA) | OK Compost (EU) | PFAS-Free | FDA Safe | Home Compost | Typical Limit* |
|---|---|---|---|---|---|---|
| Bagasse | Yes | Industrial/Home | Yes | Yes | Strong | ≤ 0.8mm |
| Aqueous Paper | Yes | Industrial/Recyclable | Yes | Yes | Varies | ≤ 1.0mm |
| PLA / CPLA | Yes | Industrial Only | Yes | Yes | No | ≤ 0.05mm (Film) |
| Cornstarch (PSM) | Yes | Industrial Only | Yes | Yes | No | ≤ 0.8mm |
* Note: Thickness checkpoints are based on typical certification annexes. Individual SKU compliance must be verified against the specific certificate holder’s technical file.
2. How to Read This Matrix Correctly
Bagasse
Bagasse typically carries the strongest cross-market sustainability story because it can combine industrial compostability, possible home-compostability routes, food-contact safety, and PFAS-free positioning when properly managed. For many EU buyers, it represents the most straightforward path into premium compostable foodservice, especially for hot food, molded fiber trays, and heavy-duty takeaway packaging. The main procurement risk is not usually the material itself, but scope mismatch between the tested certificate and the final product thickness or form.
Aqueous Paper
Aqueous-coated paper is strategically important because it sits at the intersection of compostability, repulpability, and plastic-content scrutiny. In Europe, its value is not just that it can avoid conventional poly-coating routes, but that it may reduce certain recyclability and plastic-perception barriers when correctly designed. However, this category must be handled carefully because the final compliance route can differ by structure, barrier system, and end use. Buyers should therefore avoid blanket claims and instead request SKU-level evidence on coating chemistry, recyclability pathway, and compostability scope.

PLA / CPLA
PLA and CPLA are strong candidates when the requirement is clearly industrial compostability, especially of PLA cups, lids, and CPLA cutlery, where appearance or rigidity matters. Their weakness lies in overclaiming: they should not be marketed as home compostable unless there is explicit certification. In European sourcing, the safest commercial route is to position them clearly within industrial composting infrastructure and ensure the certification wording mirrors that scope.
Cornstarch (PSM)
Cornstarch-based tableware are often attractive for buyers focused on cost, but they require the most caution in certification review because formulation differences can materially affect the legal claim. “Cornstarch” alone does not guarantee home compostability, PFAS-free status, or even equivalent biodegradation behavior. This category should therefore be sourced only with formula-specific supporting documents, not by material name alone.
IV. Beyond the Logo: How to Verify Certificates
A professional sourcing decision does not stop at receiving a PDF file by email. In real procurement practice, the difference between a test report, a certificate, and a logo-use right is where many importers fail their audit. A test report may confirm that a sample passed a laboratory method on a certain date. A certificate, by contrast, is a formal third-party approval tied to a defined product scope, a certificate holder, and an active validity period. Logo usage, in many systems, is a further commercial right governed by trademark and certification licensing rules. Treating these three as interchangeable is one of the most common compliance mistakes in compostable packaging sourcing.
1. How to Verify BPI Certificates in Practice
For North American sourcing, the first control point should be the BPI public database. BPI states that its catalog is searchable and lists certified products and the companies licensed to sell them, down to the item number. This means a sourcing officer should not merely ask, “Do you have BPI?” but should verify whether the supplier, product family, and item scope are actually visible in the BPI registry. BPI also makes clear that certification is tied to its own approved process and searchable public listing, which is exactly why database verification matters more than a standalone PDF attachment.
The practical verification process should be treated as a three-step audit. First, search the certificate holder or sublicensee in BPI’s certified product catalog. Second, confirm that the listed item category and product description match the goods you are actually buying. Third, check whether the certificate is still active and whether the SKU-level scope is still covered under the current certification cycle. BPI’s own recertification guidance notes that renewal is a formal process and can begin up to one year before expiration, which means expiry dates are not administrative details—they are audit-critical.
2. How to Verify TÜV AUSTRIA / OK Compost Certificates
For Europe-facing sourcing, the equivalent control point is the TÜV AUSTRIA OK Certification database. TÜV AUSTRIA maintains an official public database of certified products, covering a wide range of certified materials and finished items. This allows buyers to verify whether a supplier’s claim to OK compost HOME, OK compost INDUSTRIAL, or related certification is actually backed by a live product entry in the official registry.
This matters because in Europe, a sustainability claim often has two layers: the technical layer and the trademark layer. A supplier may show a laboratory result or internal declaration, but if the product is not present in an official certification system, the buyer has weak legal footing when challenged by customs, retail compliance teams, or sustainability auditors. For products carrying or referencing the Seedling route, buyers should also understand that the logo is linked to formal EN 13432-based certification through approved certification bodies, not to generic biodegradability language.
3. Validity Check: Expiry Dates, Scope, and Product Matching
Certificate verification should always include an expiry-date check, a scope check, and a specification check. A certificate can be genuine and still be commercially useless if it has expired, if it covers a different item type, or if it was issued for another manufacturer in the same supply chain without a valid authorization chain. In operational terms, buyers should compare the product ordered against the exact product family described in the certification appendix, the manufacturer name in the database, and the shipping documentation used for the transaction. If the certificate holder, the factory name, and the shipper on the Bill of Lading do not align—or cannot be linked through a documented authorization chain—the compliance file is incomplete from an audit perspective.
4. Test Report vs. Certificate: Why Both Matter
A strong sourcing file typically requires both a certificate and a lot-relevant technical record. The certificate proves that the product system has passed the formal third-party route. The supporting test documentation proves that the actual production batch or formulation remains consistent with the certified product family. This distinction becomes especially important for PFAS-free compliance, thickness-sensitive compostability claims, and private-label transactions where one company manufactures and another markets. In practice, the safest procurement approach is to require three layers of evidence:
An active certificate,
The official database listing and validation,
A recent laboratory report or compliance dossier relevant to the actual goods shipped.
That is the difference between “having paperwork” and having a defensible compliance position.
V. Strategic Sourcing: Minimizing Regulatory Risks
In mature procurement environments, certification should not be treated as a decorative sustainability feature. It is a risk-control instrument. The right certification architecture can reduce border delays, simplify retailer onboarding, strengthen supplier scoring, and improve eligibility in sustainability-driven procurement environments. The wrong architecture—especially when built on expired certificates, unmatched SKUs, or weak PFAS evidence—can create the opposite outcome: rejected shipments, relabelling costs, retailer disputes, and legal exposure.
1. Using Certification to Reduce Commercial Friction
A rigorous sourcing strategy should assume that certification reduces commercial friction before it reduces cost. In other words, the first value of certification is not usually a direct tariff reduction; it is smoother market access. A BPI-listed product is easier to defend in North American compostability conversations. A TÜV-certified and properly documented item is easier to position in the EU. An officially recognized ecolabel or equivalent science-based compliance system can also simplify procurement review in public or institutional buying environments, because these systems give buyers a structured basis for environmental decision-making. The European Commission’s green procurement guidance explicitly notes that science-based labels can help streamline procurement processes and support environmental objectives.
That is why professional buyers increasingly ask not only for a product quotation, but for a compliance dossier. In many cases, the commercial gain comes from passing internal supplier approval faster, not from negotiating a lower customs duty line. A supplier like Bioleader®, able to package certifications, food-contact support, PFAS evidence, and product-scope clarity into one sourcing file becomes materially easier to approve.
2. Sub-License Strategy as a Cost-Control Tool
For emerging brands and private-label importers, one of the most practical compliance strategies is sub-licensing, rather than starting from zero with a full independent certification cycle. BPI’s own sublicensing framework confirms that sublicensees can be linked into the certified product catalog and searchable database under the appropriate agreement structure. This is not a theoretical convenience; it is an operational shortcut. It allows growing brands to reach legal logo-usage and market-facing compostability claims faster than if they built an independent testing and certification program from the ground up.
In commercial terms, this matters because a small brand often does not need a new scientific theory—it needs a faster compliant market entry path. A supplier with an established certification portfolio can, where the certification rules and contractual structure allow, reduce the time, paperwork burden, and duplicated testing cost for private-label partners. This is where a compliance-capable manufacturer creates measurable value: by turning certification from a fixed barrier into a managed entry route.
3. Ongoing Third-Party Testing as a Supply-Chain Discipline
Long-term compliance is not maintained by one certificate alone. It is maintained by discipline over time. Best-practice sourcing requires periodic third-party testing, controlled formulation management, and consistency between certified scope and shipped goods. In practical terms, that means buyers should prefer suppliers who can support periodic verification through recognized third-party laboratories and maintain internal product-control routines around coating systems, thickness, product codes, and lot traceability.
For a company like Bioleader®, the most credible positioning is therefore not merely “we have certificates,” but “we operate a compliance maintenance model.” That model should include periodic external testing when needed, active certificate tracking, product-to-certificate mapping, and support documentation that remains usable during customer audits or retailer onboarding. The commercial value of that system is continuity: the buyer reduces the chance that a compliant SKU in Q1 becomes an exposed SKU in Q4 because of unnoticed formulation drift, expired documentation, or certificate-scope mismatch.
4. Risk Minimization in Real Procurement Terms
The practical sourcing rule is simple:
Buy the product, but audit the evidence chain.
That means checking:
whether the certification is active,
whether the test method is legally persuasive,
whether the product thickness stays inside the certified envelope,
whether the shipper and certificate holder can be linked,
and whether the market claim on pack matches the actual certification scope.
A supplier that can answer all five of those questions with evidence, not slogans, is not simply a packaging vendor. It is a lower-risk sourcing partner.
VI. 2026+ Regulatory Roadmap: What’s Next?
The next phase of compliance is not only about compostability. It is increasingly about system compatibility, recyclability, and recycled content performance. In the EU, the Packaging and Packaging Waste Regulation (PPWR), Regulation (EU) 2025/40, has moved packaging compliance into a broader operational framework. According to the European Commission, the PPWR is designed to make all packaging on the EU market recyclable in an economically viable way by 2030 and to increase the use of recycled plastics in packaging, with plastic packaging subject to minimum recycled-content requirements that rise in 2030 and 2040.
For sourcing teams, this means one important thing: compostability alone will not be enough to future-proof every packaging line. A product may be compostable today and still become strategically weak tomorrow if it cannot fit into the recyclability, recycled-content, or packaging-minimisation logic being built into the next generation of EU regulation. That does not reduce the value of compostable products; it changes the sourcing question. Buyers now need to ask not only, “Is this certified?” but also, “Is this material platform aligned with where regulation is heading over the next three to five years?”
1. PCR: Why It Matters Even if Your Current SKU Is Not a Recycled-Plastic Pack
The term PCR (Post-Consumer Recycled content) is becoming a strategic planning issue because PPWR pushes the market toward minimum recycled content in plastic packaging categories over time. Even for buyers currently focused on fiber-based or compostable formats, PCR matters for two reasons. First, many portfolios are mixed portfolios: cups, lids, liners, windows, trays, and overcaps do not always sit in the same material family. Second, procurement teams will increasingly be assessed on portfolio-wide packaging direction, not on one isolated SKU.
This means that a modern sourcing roadmap should classify packaging not only by current certification, but by future regulatory exposure. Fiber, aqueous-coated paper, compostable bioplastics, and recycled-plastic systems may all continue to have a role—but they should be sourced with a clear understanding of which legal pressures are likely to intensify first.
2. From “Certified Now” to “Defensible Later”
The most resilient buyers will be those who build a two-layer compliance strategy:
Layer 1: Current market-access compliance — BPI, TÜV, PFAS-free support, food-contact safety, logo rights.
Layer 2: Forward compatibility — recyclability logic, recycled-content readiness, minimisation requirements, and public-procurement alignment.
This is the real lesson of the post-2025 regulatory environment. Winning packaging strategies are no longer built only around one claim such as “compostable” or “plastic-free.” They are built around documentation architecture and material-platform flexibility.
Conclusion: Compliance as a Competitive Asset
The global eco-packaging market is moving into a phase where certification is no longer a badge—it is infrastructure. Buyers who treat compliance as an afterthought will continue to face hidden costs: customs delays, relabelling exposure, audit failures, and weakened retailer trust. Buyers who treat compliance as part of their core sourcing strategy will move faster, defend their claims more effectively, and build more resilient supply chains.
For that reason, the right partner is not merely the supplier with a low price. It is the supplier that can help you connect certificate validity, test-method credibility, product-scope accuracy, and future regulatory readiness into one coherent sourcing framework. That is where companies such as Bioleader can create strategic value: by supporting customers not just with packaging formats, but with a more disciplined path through the global compliance maze.
Call to Action
For Enterprise Buyers
Looking to de-risk your global supply chain? Speak with our compliance team to review your current certification chain and identify hidden documentation gaps.
For Growing Brands
Starting with sustainable packaging? Request our Practical Certification Checklist to ensure every dollar you spend is legally defensible and market-ready.
*Bioleader provides end-to-end certification support for North American and European markets.
Copyright Notice:
© 2026 Bioleader®. If you wish to reproduce or reference this content, you must provide the original link and credit the source. Any unauthorized copying will be considered an infringement.




